Preventing
Severe Preeclampsia
1st and 2nd Trimester
Uterine Artery
Measurements
Preeclampsia-The Problem
Globally, preeclampsia occurs in 2 to 8% of pregnancies. Hypertensive disease in pregnancy accounts for 26% of maternal deaths in Latin America and the Caribbean, while in Africa and Asia only 9%. In the United States the rate of preeclampsia increased 25% from 1987 to 2004, with almost a 7-fold increase in severe preeclampsia. The cost in the United States for a 12-month period has been estimated be $2.18 billion for women and $1.15 billion for treatment of infants born to mothers with preeclampsia.
Women with severe preeclampsia have a 20-fold increased of maternal mortality than if preeclampsia in a less sever form is present. Currently, there are two approaches to identify patients at increased risk for severe preeclampsia. The first is using historical data, and the second is using diagnostic ultrasound to evaluate the uterine artery blood flow. These options will be discussed below as both approaches have demonstrated a decrease in severe preeclampsia when treating patients with low-dose aspirin prior to 16 weeks of pregnanccy.
While there are RISK FACTORS for Preeclampsia,
IT OFTEN OCCURS DURING THE 1ST PREGNANCY WITHOUT ANY WARNING!
RISK FACTORS FOR DEVELOPING PREECLAMPSIA
• 1st Pregnancy
• Multiple gestations
• Preeclampsia in a previous pregnancy
• Chronic hypertension
• Diabetes prior to current pregnancy
• Diabetes diagnosed during the current pregnancy
• Thrombophilia
• Systemic lupus erythematosus
• Pre-pregnancy body mass index greater than 30
• Antiphospholipid antibody syndrome
• Maternal age 35 years and older
• Kidney disease
• In-vitro Fertilization
• Obstructive sleep apnea
Mild Preeclampsia-Diagnosis
Pregnant Women with No Previous History of Preeclampsia or Chronic Hypertension
Hypertension
Systolic blood pressure >140 mmHg or diastolic blood pressure >90 mmHg identified on 2 occasions 4 hours apart after 20 weeks of pregnancy in a patient with no previous history of preeclampsia and the new onset of 1 or more of the following laboratory findings:
• Protein in the urine of more than 300 mg in a 24-hour urine collection
• Protein in the urine on a dipstick of 2+ or greater
• Urine protein/creatinine ratio > 0.3 mg/mg or 30 mg/mmol
Pregnant Woman with Previous Hypertension
Definition: New onset of protein in the urine (see above) or other symptoms after 20 weeks of pregnancy
Pregnant Women with Previous Hypertension and Protein in the Urine
Definition: Worsening of the hypertension in the 3rd trimester of pregnancy with any of the above physical findings.
Severe Preeclampsia-Diagnosis
Definition: Systolic blood pressure greater than 160 mmHg and/or diastolic blood pressure greater than 110 mmHg with one or more of the following:
Laboratory Findings
• Platelet count of <100,000/microL
• Serum creatinine >1.1 mg/dl
• Liver transaminases at least 2 times greater than the normal laboratory values
• Pulmonary edema
Physical Findings
• New onset of a headache not response to traditional treatment
• Cerebral or visual symptoms, blurred vision, flashing lights, appearance of blind spots or loss of vision
• Pain in the right upper abdomen
Adapted from: American College of Obstetricians and Gynecologists (ACOG) Practice Bulletin No. 202: Gestational Hypertension and Preeclampsia. Obstet Gynecol 2019; 133:e1-e25
Associated with High Risk
for Maternal and Fetal
Complications
Identification of High-Risk Patients for Preeclampsia Using Historical Information
In 2018 the American College of Obstetricians has endorsed recommendations from the U.S. Preventive Services Task Force in 2014 that identified risk factors that placed a patient at increased risk for preeclampsia. Patients who are identified by either 1 or 2 risk factors (see Table below) would qualify to received low-dose aspirin to prevent preeclampsia. However, these risk factors do not identify low-risk pregnancies that make up the majority who develop preeclampsia during the course of their pregnancy. To identify these low-risk patients, uterine artery Doppler evaluation should be considered (see section below).
American College of Obstetrics and Gynecology
(2018)
Factors to Identify Patients At Risk for Severe Preeclampsia
High Risk for Preeclampsia
• History of preeclampsia, especially when accompanied by an adverse outcome
• Multifetal gestation
• Chronic hypertension
• Type 1 or 2 diabetes
• Renal disease
• Autoimmune disease (systemic lupus erythematosus, antiphospholipid syndrome
Moderate Risk for Preeclampsia
Increaesd Risk if the patient has TWO or more of the following risk factors:
• 1st Pregnancy
• Obesity (body mass index >30) (See Calculator)
• Family history of preeclampsia (mother or sister)
• Sociodemographic characteristics (African American race, low socioeconomic status)
• Age 35 year or older
• Personal history (low birth weight or small for gestational age previous pregnancy, adverse pregnancy outcome, more than 10-year pregnancy interval)
LeFevre ML; U.S. Preventive Services Task Force. Low-dose aspirin use for the prevention of morbidity and mortality from preeclampsia: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2014;161(11):819–826. doi:10.7326/M14-1884
A recent study using historical criteria suggested by the American College of Obstetricians and Gynecologists (ACOG) only identified 54.6% of patients who developed preeclampsia with a false-positive rate of 20.4%
American Journal of Obstetrics and Gynecology, December, 2019

Read the Study
Is There A Role for Ultrasound to Identify Low-Risk Patients Who Could Develop Severe Preeclampsia?
While historic factors identify women at risk for preeclampsia, as described above, the majority of women who develop this disease HAVE NO RISK FACTORS.
An important study was published in the Canadian Medical Association Journal in 2008, followed by another large study from Europe in 2014, have demonstrated the value of Doppler evaluation of the uterine arteries performed during the 1st trimester of pregnancy (11 to 14 weeks) to identify LOW RISK women who are at increased risk for severe preeclampsia.
The study in 2014 compared detection rates using historical factors vs abnormal uterine artery Doppler in 55,974 low risk patients and found the following:
- The detection rate for severe preeclampsia using uterine artery Doppler in low risk patients was 47.8% with a false-positive rate of 7.9%. Therefore, to prevent one case of severe preeclampsia, 173 women would have to be treated with low-dose aspirin.
- Conversely, using any of the historical risk factors the number of patients needed to be treated to prevent 1 case of severe preeclampsia varied between 1 in 256 to 1 in 1,429.
- Screening all patients using uterine artery Doppler also resulted in identifying patients at risk for the following:
-
- Early growth restriction (see section on the website describing this problem) 39.2%, with a false-positive rate of 6.9%.
- Stillbirth with a sensitivity of 14.5% and a false-positive rate of 8.7%.
-
- An abnormal Doppler of the uterine artery obtained in low risk patients had a similar screening performance as high-risk patients.
Conclusions:
- Patients with no risk factors for preeclampsia, which makes up the majority of pregnant women, especially those having their first baby, have NO options for detecting their risk if historical factors are only used.
- Using Doppler evaluation of the uterine arteries in low risk patients detects as many patients who develop severe preeclampsia as those who have high-risk historical factors
- 1st trimester Doppler assessment of the uterine arteries to identify high-risk fetuses should be offered to all patients, irrespective of their risk using historical factors because abnormal values also identify other fetal conditions such as abnormal growth, as well as risks for a stillbirth.

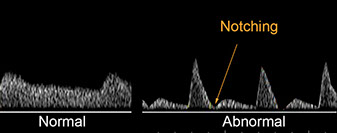
The red overlay demonstrates the normal shape of the Doppler waveform of the uterine artery. The images to the right demonstrate a "notch" that is abnormal. The notch suggests that the vessels in the placenta have not developed properly, thus resulting in an increased resistance to blood flow.
Identification Low-Risk Patients by Historical Criteria Who Have a High Risk for Preeclampsia Following Doppler Evaluation of the Uterine Arteries
Treatment Using Low-Dose Aspirin
Low-dose aspirin is the recommended treatment for patients at risk for preeclampsia. Researchers have recommended varying doses of aspirin ranging between 60 mg/day to 160 mg/day.
Since low-dose aspirin only is available in 81 mg tablets. Therefore, the physican may prescribe either one 81 mg tablet or two 81 milligram tablets to be taken after the evening meal.
At the fetal diagnostic the protocol is as follows:
1st Trimester Evaluation of the Uterine Arteries (weeks 11 to 14)
- Take two 81 mg tablets (162 milligrams) in the evening.
2nd Trimester Evaluation of the Uterine Arteries (weeks 16 to 22)
- If “notching persists into the 2nd trimester, the above regimen is continued until 36 weeks.
- If “notching has resolved, and is no longer present, then the aspirin is stopped.
- If “notching is detected then the aspirin regimen used in the 1st trimester is continued until 36 weeks.
- If “notching is no longer present, then the above aspirin regimen is continued.
- If the patient was not evaluated during the 1st trimester, but is noted to have abnormal Doppler of the uterine arteries, then the regimen is as follows:
-
- Take two 81 mg tablets (162 milligrams) in the evening.



